Nomenclature
What is nomenclature?
Nomenclature is another word for naming molecules. When naming a molecule, there are specific rules. This section will review these rules.
The first thing you should do when trying to name a compound is determining whether the first element in the compound is a metal or a non-metal. If the compound starts with a metal and is attached to a non-metal, it is an ionic
compound. The rule for this would be to write the first element’s name and then the second element along with ‘ide’.
Examples:
NaCl – Sodium chloride
K2O – Potassium oxide
AlCl3 – Aluminum chloride
If the elements are both non-metals then this is called a covalent bond.
When naming a covalent bond, you write the first elements name then the second
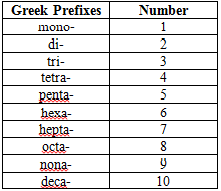
elements name along with the word ‘ide’.Along with this, you must use Greek
prefixes like mono, di, and tri. If the second element contained three atoms, it
would have ‘tri’ in front of its name. Remember, when the first element has one
atom, don’t use the word mono. However, use Greek prefixes in all other
cases.
Examples:
NF3 – Nitrogen trifluoride
CO2 – Carbon dioxide
PCl5 – Phosphorous pentachloride
N2O4 - Dinitrogen tetroxide
Nomenclature is another word for naming molecules. When naming a molecule, there are specific rules. This section will review these rules.
The first thing you should do when trying to name a compound is determining whether the first element in the compound is a metal or a non-metal. If the compound starts with a metal and is attached to a non-metal, it is an ionic
compound. The rule for this would be to write the first element’s name and then the second element along with ‘ide’.
Examples:
NaCl – Sodium chloride
K2O – Potassium oxide
AlCl3 – Aluminum chloride
If the elements are both non-metals then this is called a covalent bond.
When naming a covalent bond, you write the first elements name then the second
elements name along with the word ‘ide’.Along with this, you must use Greek
prefixes like mono, di, and tri. If the second element contained three atoms, it
would have ‘tri’ in front of its name. Remember, when the first element has one
atom, don’t use the word mono. However, use Greek prefixes in all other
cases.
Examples:
NF3 – Nitrogen trifluoride
CO2 – Carbon dioxide
PCl5 – Phosphorous pentachloride
N2O4 - Dinitrogen tetroxide
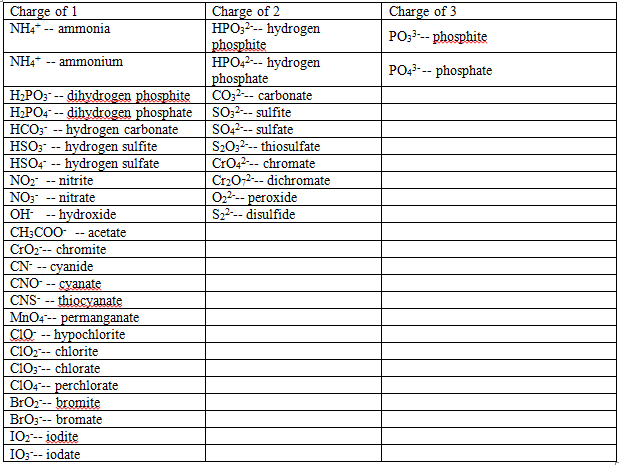
If the elements are a metal and a polyatomic ion, you would simply write the name of the first element and the name of the polyatomic ion.
Examples:
Na3PO4 – Sodium phosphate
AgNO3 – Silver nitrate
CaCO3 – Calcium carbonate
Examples:
Na3PO4 – Sodium phosphate
AgNO3 – Silver nitrate
CaCO3 – Calcium carbonate
When there is a metalloid, you would treat it as if it were a non-metal, using the same rules.
Examples:
BCl3 – Boron trichloride
SiO – Silicon monoxide
BF3 – Boron trifluoride
Examples:
BCl3 – Boron trichloride
SiO – Silicon monoxide
BF3 – Boron trifluoride
When there is a transition metal involved, the only difference is that you would have to include the charge in brackets next to the name of the transition metal.
Examples:
CrCl3 – Chromium (III) chloride
Cu2O – Copper (I) oxide
FeO – Iron (II) oxide
However, when there is a transition metal, you must also consider the placement of the transition metal on the periodic table. There is a cluster of transition metals that form the shape of a right angle triangle. The elements included are
Aluminum, Galium, Indium, Zinc, Cadmium, and Silver. These six transition metals only have one possible option for a charge therefore you do not have to specify their charge when writing their name. All other transition metals have at least two possible charges.
Examples:
Al2S3 – Aluminum sulfide
GaN - Gallium nitride
AgNO3 - Silver nitrate
The last rule is more of a memorization than a rule because there are some compounds that are acids. When a compound is an acid, it is simply a matter or memorizing their names.
Here is a list of the most common acids that you would be expected to know:
HF – Hydrofluoric acid
HCl – Hydrochloric acid
HBr – Hydrobromic acid
HI – Hydroiodic acid
H2S – Hydrosulfuric acid
HNO3 – Nitric acid
H2SO4 - Sulfuric acid
CH3COOH – Acetic acid
Practice
Name the following compounds:
1) NaNO3
2) CuBr2
3) MgO
4) BaSO3
5) HCl
6) PbO
7) Li3PO4
8) NH4OH
9) KCN
10) FeO
11) Cr(NO3)2
Answers:
1) Sodium nitrate
2) Copper (II) bromide
3) Magnesium oxide
4) Barium sulfate
5) Hydrochloric acid
6) Lead (II) oxide
7) Lithium phosphate
8) Ammonium hydroxide
9) Potassium cyanide
10) Iron (II) oxide
11) Chromium (II) nitrate
Write the compounds for the following names:
1) Ammonium sulphide
2) Magnesium phosphate
3) Iron (II) phosphate
4) Calcium sulfite
5) Aluminum nitrate
6) Iron (II) chloride
7) Sodium chloride
8) Copper (II) hydroxide
9) Calcium bromide
10) Calcium fluoride
11) Acetic Acid
Answers:
1) (NH4)2S
2) Mg3(PO4)2
3) Fe3(PO4)2
4) CaSO3
5) Al(NO3)3
6) FeCl
7) NaCl
8) Cu(OH)2
9) CaBr2
10) CaF2
11) CH3COOH
Here is a list of extra practice worksheets:
http://chemistry.about.com/od/nomenclature/a/nomenclature-ionic-compounds.htm
http://www.shodor.org/unchem/basic/nomen/index.html
http://www.emsb.qc.ca/laurenhill/science/name.html
http://dkreutz.basd.k12.wi.us/A10-bonds.html
http://www.woodrow.org/teachers/chemistry/links/chem1/namingcomp.html
http://www.fernbank.edu/Chemistry/nomen.html
http://preparatorychemistry.com/Bishop_nomenclature_help.htm