Precipitate reactions
Solutions and Solubility
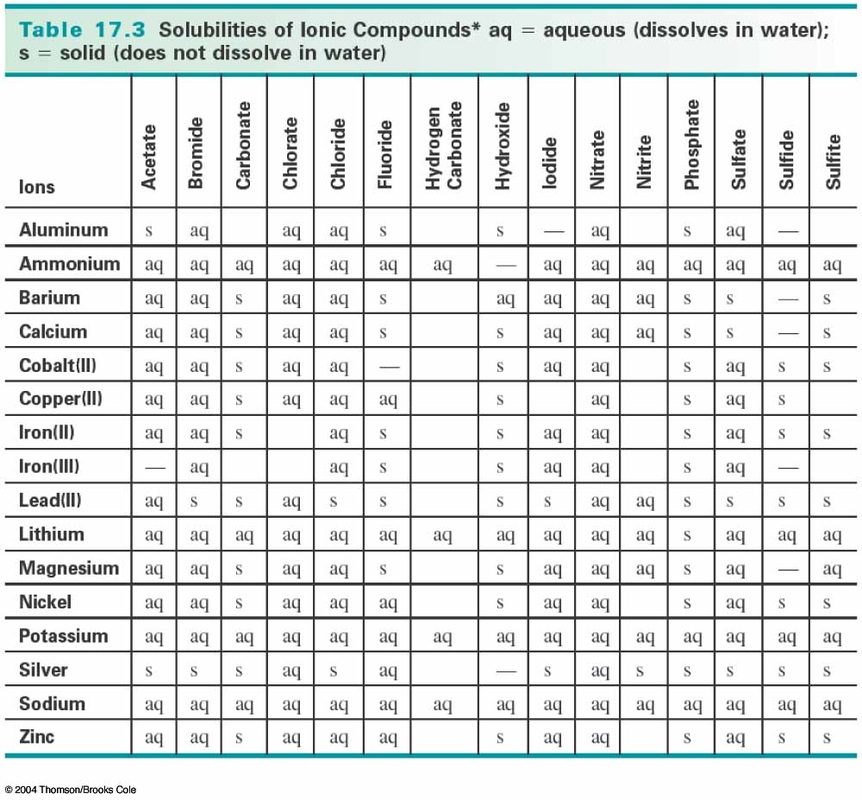
In order to be considered soluble, a substance must have a concentration of at least 0.1mol/L at room temperature (25°). Any substance with a concentration less than this value is insoluble.
*Important: All compounds with group 1A ions, NO3- ions and NH4+ ions are soluble (no precipitate formed).
A list of the rules regarding solubility in water can be found here:
http://www.files.chem.vt.edu/RVGS/ACT/notes/solubility_rules.html
The chart below can also be used to determine which compounds are soluble and which are not.
In order to be considered soluble, a substance must have a concentration of at least 0.1mol/L at room temperature (25°). Any substance with a concentration less than this value is insoluble.
*Important: All compounds with group 1A ions, NO3- ions and NH4+ ions are soluble (no precipitate formed).
A list of the rules regarding solubility in water can be found here:
http://www.files.chem.vt.edu/RVGS/ACT/notes/solubility_rules.html
The chart below can also be used to determine which compounds are soluble and which are not.
When two or more substances react with one another (at least one of them being an electrolytic solution) and a solid is formed, a precipitate reaction has taken place. The solid formed is called the precipitate.
Total and Net Ionic Equations
If a substance falls into one of the following categories:
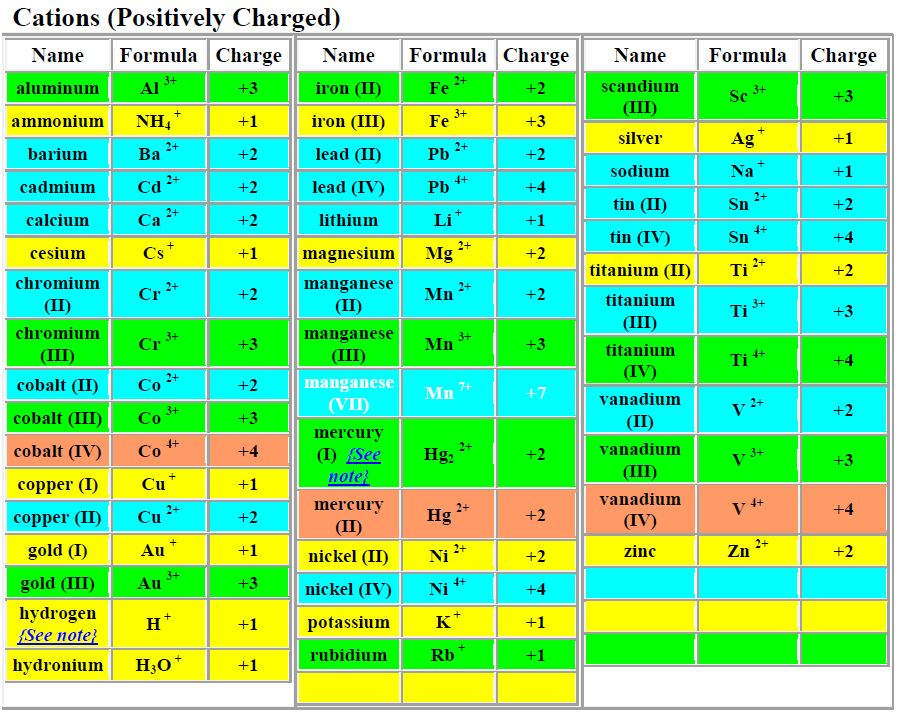
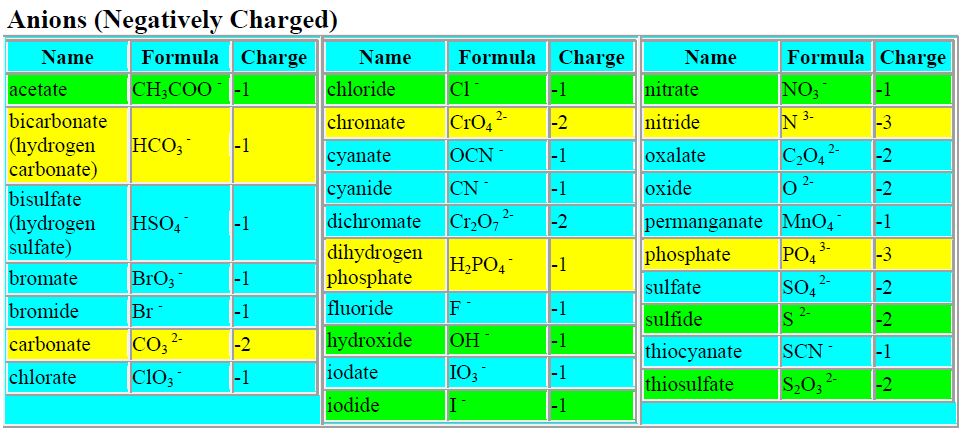
1) Ionic compound (metal-nonmetal)
2) Strong acid
3) Strong base (all with the exception of NH4OH)
then they break up into their component ions upon dissolution.
Strong acids include:
-HCl
-HNO3
-HClO4
-H2SO4
-H3PO4 (moderately strong)
Covalent substances are not broken up into ions in the equation because they are not ionized. Precipitates and substances in solid or gaseous states are not broken up into ions because they are not in solution.
*H2CO3 (a weak acid) will break apart into H2O(l) and CO2(g) upon formation. NH4OH (a weak base) will do the same, decomposing into NH3(g) andH2O(l).
If a substance falls into one of the following categories:
1) Ionic compound (metal-nonmetal)
2) Strong acid
3) Strong base (all with the exception of NH4OH)
then they break up into their component ions upon dissolution.
Strong acids include:
-HCl
-HNO3
-HClO4
-H2SO4
-H3PO4 (moderately strong)
Covalent substances are not broken up into ions in the equation because they are not ionized. Precipitates and substances in solid or gaseous states are not broken up into ions because they are not in solution.
*H2CO3 (a weak acid) will break apart into H2O(l) and CO2(g) upon formation. NH4OH (a weak base) will do the same, decomposing into NH3(g) andH2O(l).
Writing a net ionic equation
A more detailed explanation of net ionic equations can be found here:
http://www.occc.edu/kmbailey/chem1115tutorials/Net_Ionic_Eqns.htm
Example 1
Given equation: (NH4)2SO4(aq) + BaCl2(aq) ---> BaSO4(s) + 2NH4Cl(aq)
Total ionic equation: 2NH4+(aq) + SO4 2-(aq) + Ba2+(aq) + 2Cl- (aq) ---> BaSO4(s) + 2NH4+(aq) + 2Cl- (aq)
Since BaSO4 is a precipitate, it does not need to be split up into ions. Also, any subscripts that the substance has must be brought out to the front of it.
Net ionic equation: SO4 2-(aq) + Ba2 +(aq) ---> BaSO4(s)
You may have noticed that there are ions common to both sides of the equation. These are called spectator ions, and do not take part in the reaction. They can therefore be crossed out from the net ionic equation.
Example 2
Given equation: CaCO3(s) + 2HNO3 +(aq) ---> Ca(NO3)2(aq) + H2O(l) + CO2(g)
Total ionic equation: CaCO3(s) + 2H +(aq) + 2NO3 -(aq) ---> Ca2 +(aq) + 2NO3 -(aq) + H2O(l) +CO2(g)
Since CaCO3 is a solid reagent, it does not split up into ions.
Net ionic equation: CaCO3(s) + 2H +(aq) ---> H2O(l) + CO2(g)
Calcium carbonate reacts with hydrogen to form water and carbon dioxide.
http://www.occc.edu/kmbailey/chem1115tutorials/Net_Ionic_Eqns.htm
Example 1
Given equation: (NH4)2SO4(aq) + BaCl2(aq) ---> BaSO4(s) + 2NH4Cl(aq)
Total ionic equation: 2NH4+(aq) + SO4 2-(aq) + Ba2+(aq) + 2Cl- (aq) ---> BaSO4(s) + 2NH4+(aq) + 2Cl- (aq)
Since BaSO4 is a precipitate, it does not need to be split up into ions. Also, any subscripts that the substance has must be brought out to the front of it.
Net ionic equation: SO4 2-(aq) + Ba2 +(aq) ---> BaSO4(s)
You may have noticed that there are ions common to both sides of the equation. These are called spectator ions, and do not take part in the reaction. They can therefore be crossed out from the net ionic equation.
Example 2
Given equation: CaCO3(s) + 2HNO3 +(aq) ---> Ca(NO3)2(aq) + H2O(l) + CO2(g)
Total ionic equation: CaCO3(s) + 2H +(aq) + 2NO3 -(aq) ---> Ca2 +(aq) + 2NO3 -(aq) + H2O(l) +CO2(g)
Since CaCO3 is a solid reagent, it does not split up into ions.
Net ionic equation: CaCO3(s) + 2H +(aq) ---> H2O(l) + CO2(g)
Calcium carbonate reacts with hydrogen to form water and carbon dioxide.
Extra Notes and Practice
http://www.files.chem.vt.edu/RVGS/ACT/notes/Notes_on_Net_ionic_rxns.html
http://pages.towson.edu/ladon/PracticeNIE.htm
http://pages.towson.edu/ladon/PracticeNIEAnswers.htm (answers to previous)
http://www.wjmouat.com/Teachers/BHutchinson/Chemistry/Chem11/Unit%206/PS64.htm
http://www.chem.wisc.edu/deptfiles/genchem/sstutorial/Text11/NetIonic/NetPractice.htm
Labs
http://www.jdchs.org/pdfs/chemlabs/7_Precipitation.pdf
http://swc2.hccs.edu/pahlavan/intro_labs/Exp_10_Precipitation_Reactions_(Metathesis_Reactions).pdf
http://hrsbstaff.ednet.ns.ca/benoitn/chem12/solutions/lab_precipitation_reactions.htm
http://staff.fcps.net/jswango/unit4/reactions/PRECIPITATION%20REACTION%20LAB.pdf
Videos
http://www.youtube.com/watch?v=dXqelH1MxGw
http://www.youtube.com/watch?v=pCpzhf8hNTc
http://www.youtube.com/watch?v=2B-K6BYxr-s
http://www.files.chem.vt.edu/RVGS/ACT/notes/Notes_on_Net_ionic_rxns.html
http://pages.towson.edu/ladon/PracticeNIE.htm
http://pages.towson.edu/ladon/PracticeNIEAnswers.htm (answers to previous)
http://www.wjmouat.com/Teachers/BHutchinson/Chemistry/Chem11/Unit%206/PS64.htm
http://www.chem.wisc.edu/deptfiles/genchem/sstutorial/Text11/NetIonic/NetPractice.htm
Labs
http://www.jdchs.org/pdfs/chemlabs/7_Precipitation.pdf
http://swc2.hccs.edu/pahlavan/intro_labs/Exp_10_Precipitation_Reactions_(Metathesis_Reactions).pdf
http://hrsbstaff.ednet.ns.ca/benoitn/chem12/solutions/lab_precipitation_reactions.htm
http://staff.fcps.net/jswango/unit4/reactions/PRECIPITATION%20REACTION%20LAB.pdf
Videos
http://www.youtube.com/watch?v=dXqelH1MxGw
http://www.youtube.com/watch?v=pCpzhf8hNTc
http://www.youtube.com/watch?v=2B-K6BYxr-s