stoichiometry
Stoichiometry is the measuring of the quantity of the different substances involved in a chemical
reaction. The calculations used in stoichiometry are in accordance to the law of conservation of matter – matter can neither be created nor destroyed.
reaction. The calculations used in stoichiometry are in accordance to the law of conservation of matter – matter can neither be created nor destroyed.
Mole
The mole is a unit that is used to indicate the amount of particles, atoms, molecules, etc. Since these are too numerous to count individually, moles are used instead. Just like a pair of socks means two socks and a dozen eggs is equivalent to twelve eggs, one mole is equal to 6.02 ×1023 particles. This quantity corresponds to Avogadro’s number
(NA), which is 6.02 ×1023. The mole is represented by the symbol mol.
The mole is a unit that is used to indicate the amount of particles, atoms, molecules, etc. Since these are too numerous to count individually, moles are used instead. Just like a pair of socks means two socks and a dozen eggs is equivalent to twelve eggs, one mole is equal to 6.02 ×1023 particles. This quantity corresponds to Avogadro’s number
(NA), which is 6.02 ×1023. The mole is represented by the symbol mol.
Molar Mass
The molar mass of a substance – element or compound – refers to how much a mole of that substance weighs. It is equivalent to either the atomic mass of the element or to the combined atomic masses of the elements that form the compound (depending on the nature of the substance), and is measured in g/mol (grams per mole).
Example 1: What is the molar mass of Na?
The atomic weight of Na is 23 amu (atomic mass units).
The molar mass of Na is therefore 23g/mol.
Example 2: What is the molar mass of H2O?
The atomic weight of H is 1 amu ---> molar mass is 1g/mol.
The atomic weight of O is 16 amu ---> molar mass is 16g/mol.
Since there are 2 hydrogen atoms in one molecule of water, the molar mass of each atom must be included in the calculation.
1g × 2 + 16g = 18g
mol mol mol
The molar mass of water is 18g/mol.
The molar mass of a substance – element or compound – refers to how much a mole of that substance weighs. It is equivalent to either the atomic mass of the element or to the combined atomic masses of the elements that form the compound (depending on the nature of the substance), and is measured in g/mol (grams per mole).
Example 1: What is the molar mass of Na?
The atomic weight of Na is 23 amu (atomic mass units).
The molar mass of Na is therefore 23g/mol.
Example 2: What is the molar mass of H2O?
The atomic weight of H is 1 amu ---> molar mass is 1g/mol.
The atomic weight of O is 16 amu ---> molar mass is 16g/mol.
Since there are 2 hydrogen atoms in one molecule of water, the molar mass of each atom must be included in the calculation.
1g × 2 + 16g = 18g
mol mol mol
The molar mass of water is 18g/mol.
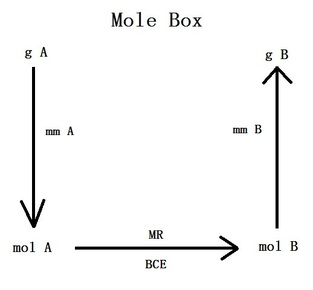
Mole Box
The mole box is used to convert the measurements of one substance to the measurements of another substance when both are involved in a chemical reaction. You may start from any part of the box and make your way to the solution.
Stoichiometry Problems
Before starting any problem, you must have a balanced chemical equation.
For example:
H2O (l) + elec E ---> H2(g) + O2(g)
When this equation is balanced, it becomes
2H2O (l) + elec E --->2H2(g) + O2(g)
This will tell us the ratios between the amounts of the different chemicals.
Using the balanced chemical equation, we can see that when 2 moles of water are decomposed, 2 moles of hydrogen and 1 mole of oxygen are produced. The ratios are as follows:
2 mol H2O : 2 mol H2 or 2 mol H2 : 2 mol H2O*
2 mol H2O : 1 mol O2 or 1 mol O2 : 2 mol H2O*
2 mol H2 : 1 mol O2 or 1 mol O2 : 2 mol H2*
*The order of the values in the ratio does not matter.
Stoichiometry problems can be solved in one step, two steps or three steps with the use of dimensional analysis.
Using the previous balanced chemical equation:
One-Step Problem – How many moles of oxygen can be made with 4 moles of water?
4 mol H2O ---> ? mol O2
mol A ---> mol B
4 mol H2O × 1 mol O2 = 2 mol O2
2 mol H2O
2.5 mol of oxygen can be made.
Two-Step Problem – How many grams of oxygen can I make with 30 moles of hydrogen?
30 mol H2 ---> ? g O2
mol A ---> g B
30 mol H2 × 1mol O2 × 32g O2 = 480g O2
2 mol H2 1 mol O2
I can make 480g of oxygen.
Three-Step Problem – Using 175g of water, how many grams of oxygen can be produced?
175g H2O ---> ? g O2
g A ---> g B
175g H2O × 1 mol H2O × 1 mol O2 × 32g O2 = 156g O2
18g H2O 2 mol H2O 1 mol O2
156g O2 can be produced.
Before starting any problem, you must have a balanced chemical equation.
For example:
H2O (l) + elec E ---> H2(g) + O2(g)
When this equation is balanced, it becomes
2H2O (l) + elec E --->2H2(g) + O2(g)
This will tell us the ratios between the amounts of the different chemicals.
Using the balanced chemical equation, we can see that when 2 moles of water are decomposed, 2 moles of hydrogen and 1 mole of oxygen are produced. The ratios are as follows:
2 mol H2O : 2 mol H2 or 2 mol H2 : 2 mol H2O*
2 mol H2O : 1 mol O2 or 1 mol O2 : 2 mol H2O*
2 mol H2 : 1 mol O2 or 1 mol O2 : 2 mol H2*
*The order of the values in the ratio does not matter.
Stoichiometry problems can be solved in one step, two steps or three steps with the use of dimensional analysis.
Using the previous balanced chemical equation:
One-Step Problem – How many moles of oxygen can be made with 4 moles of water?
4 mol H2O ---> ? mol O2
mol A ---> mol B
4 mol H2O × 1 mol O2 = 2 mol O2
2 mol H2O
2.5 mol of oxygen can be made.
Two-Step Problem – How many grams of oxygen can I make with 30 moles of hydrogen?
30 mol H2 ---> ? g O2
mol A ---> g B
30 mol H2 × 1mol O2 × 32g O2 = 480g O2
2 mol H2 1 mol O2
I can make 480g of oxygen.
Three-Step Problem – Using 175g of water, how many grams of oxygen can be produced?
175g H2O ---> ? g O2
g A ---> g B
175g H2O × 1 mol H2O × 1 mol O2 × 32g O2 = 156g O2
18g H2O 2 mol H2O 1 mol O2
156g O2 can be produced.
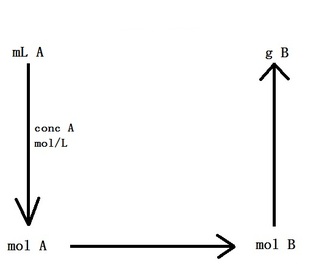
Molarity and Stoichiometry
The amount of reactants and products involved in a chemical reaction between two solutions can also be calculated in a manner similar to the questions above.
The amount of reactants and products involved in a chemical reaction between two solutions can also be calculated in a manner similar to the questions above.
The following formula can be useful for your calculations:
CV = n
where C is the concentration in mol/L (moles per litre), V is the volume in L (litres) and n is the numbers of moles.
Example:
2 NaOH(aq) + H2SO4(aq) ---> Na2SO4(aq) + 2H2O(l)
If you have 200ml of 5mol/L NaOH solution, how many grams of water will be produced?
200mL NaOH = 0.2LNaOH
0.2L NaOH × 5 mol NaOH × 2 mol H2O × 18g H2O = 18g H2O
L 2 mol NaOH 1 mol H2O
18g of water will be produced.
If I have 80.0g of water, how many litres of 3.5mol/L H2SO4 solution did I use?
80.0g H2O × 1 mol H2O × 1 mol H2SO4 × 1L H2SO4 = 0.635L H2SO4
18g H2O 2 mol H2O 3.5mol H2SO4
I used 0.635L of H2SO4 solution.
CV = n
where C is the concentration in mol/L (moles per litre), V is the volume in L (litres) and n is the numbers of moles.
Example:
2 NaOH(aq) + H2SO4(aq) ---> Na2SO4(aq) + 2H2O(l)
If you have 200ml of 5mol/L NaOH solution, how many grams of water will be produced?
200mL NaOH = 0.2LNaOH
0.2L NaOH × 5 mol NaOH × 2 mol H2O × 18g H2O = 18g H2O
L 2 mol NaOH 1 mol H2O
18g of water will be produced.
If I have 80.0g of water, how many litres of 3.5mol/L H2SO4 solution did I use?
80.0g H2O × 1 mol H2O × 1 mol H2SO4 × 1L H2SO4 = 0.635L H2SO4
18g H2O 2 mol H2O 3.5mol H2SO4
I used 0.635L of H2SO4 solution.
Extra Notes and Practice
Stoichiometry
http://www.chem4kids.com/files/react_stoichio.html
http://www.science.uwaterloo.ca/~cchieh/cact/c120/stoichio.html
Mole
http://chemistry.about.com/cs/generalchemistry/f/blmole.htm
http://misterguch.brinkster.net/oct2000.pdf
http://emp.byui.edu/PECKK/Chem105/molsheet.htm
Molar Mass
http://misterguch.brinkster.net/molarmass.html
http://chemistry.about.com/library/formulamass.pdf
http://chemistry.about.com/library/formulamassanswer.pdf (answers to previous)
http://galileoweb.org/oconnorh/2010/09/28/chemistry-molar-mass-problems/
Dimensional Analysis
http://www2.ucdsb.on.ca/tiss/stretton/Basic_skills/Dimensional_Analysis_Contents.htm
http://www.alysion.org/dimensional/fun.htm
http://westwood.sjsd.net/~dshoesmith/FOV1-0003789A/FOV1-000378A0/Dimensional%20Analysis%20Worksheet%202.doc?Plugin=Metro
http://www.petoskeyschools.org/ochs.tf.t/DIMENSIONAL%20ANALYSIS.doc
http://learnlab.hfcc.edu/sites/learnlab.hfcc.edu/files/dimanalsci.pdf
Stoichiometry Problems
http://www.sciencegeek.net/Chemistry/taters/Unit4Stoichiometry.htm
http://www.sciencebugz.com/chemistry/chprbstoich.html
http://misterguch.brinkster.net/PRA004.pdf
http://misterguch.brinkster.net/PRA048.pdf http://misterguch.brinkster.net/PRA100.doc
http://www.thegeoexchange.org/chemistry/stoichiometry/stoichiometry-problems.html
http://academic.evergreen.edu/curricular/matterandmotion/chem_phys%5Cpractice_problems.htm(first part only)
http://www.scienceiscool.org/stoichiometry/problems.html
Videos
Moles
http://www.youtube.com/watch?v=AsqEkF7hcII
http://www.youtube.com/watch?v=SjQG3rKSZUQ
Stoichiometry
http://www.youtube.com/watch?v=q3WAd2ogqlw
http://www.youtube.com/watch?v=JIw_BmFZdhI
http://www.youtube.com/watch?v=tEn0N4R2dqA
Labs
http://misterguch.brinkster.net/mlx039.doc
http://www.gulfislandssecondary.ca/curtis/Chemistry%2011_files/labset.pdf
http://haspi.org/curriculum-library/MedicalChemistry/04%20Standard%203%20Conservation%20of%20Matter%20and%20Stoichiometry/Labs%20and%20Activities/Stoichiometry%20Lab%20baking%20soda%20and%20vinegar%202.pdf
http://shelbychem.weebly.com/uploads/4/5/0/3/4503102/decomposition_of_sodium_chlorate.doc
http://shelbychem.weebly.com/uploads/4/5/0/3/4503102/lead_iodide_lab.doc
Stoichiometry
http://www.chem4kids.com/files/react_stoichio.html
http://www.science.uwaterloo.ca/~cchieh/cact/c120/stoichio.html
Mole
http://chemistry.about.com/cs/generalchemistry/f/blmole.htm
http://misterguch.brinkster.net/oct2000.pdf
http://emp.byui.edu/PECKK/Chem105/molsheet.htm
Molar Mass
http://misterguch.brinkster.net/molarmass.html
http://chemistry.about.com/library/formulamass.pdf
http://chemistry.about.com/library/formulamassanswer.pdf (answers to previous)
http://galileoweb.org/oconnorh/2010/09/28/chemistry-molar-mass-problems/
Dimensional Analysis
http://www2.ucdsb.on.ca/tiss/stretton/Basic_skills/Dimensional_Analysis_Contents.htm
http://www.alysion.org/dimensional/fun.htm
http://westwood.sjsd.net/~dshoesmith/FOV1-0003789A/FOV1-000378A0/Dimensional%20Analysis%20Worksheet%202.doc?Plugin=Metro
http://www.petoskeyschools.org/ochs.tf.t/DIMENSIONAL%20ANALYSIS.doc
http://learnlab.hfcc.edu/sites/learnlab.hfcc.edu/files/dimanalsci.pdf
Stoichiometry Problems
http://www.sciencegeek.net/Chemistry/taters/Unit4Stoichiometry.htm
http://www.sciencebugz.com/chemistry/chprbstoich.html
http://misterguch.brinkster.net/PRA004.pdf
http://misterguch.brinkster.net/PRA048.pdf http://misterguch.brinkster.net/PRA100.doc
http://www.thegeoexchange.org/chemistry/stoichiometry/stoichiometry-problems.html
http://academic.evergreen.edu/curricular/matterandmotion/chem_phys%5Cpractice_problems.htm(first part only)
http://www.scienceiscool.org/stoichiometry/problems.html
Videos
Moles
http://www.youtube.com/watch?v=AsqEkF7hcII
http://www.youtube.com/watch?v=SjQG3rKSZUQ
Stoichiometry
http://www.youtube.com/watch?v=q3WAd2ogqlw
http://www.youtube.com/watch?v=JIw_BmFZdhI
http://www.youtube.com/watch?v=tEn0N4R2dqA
Labs
http://misterguch.brinkster.net/mlx039.doc
http://www.gulfislandssecondary.ca/curtis/Chemistry%2011_files/labset.pdf
http://haspi.org/curriculum-library/MedicalChemistry/04%20Standard%203%20Conservation%20of%20Matter%20and%20Stoichiometry/Labs%20and%20Activities/Stoichiometry%20Lab%20baking%20soda%20and%20vinegar%202.pdf
http://shelbychem.weebly.com/uploads/4/5/0/3/4503102/decomposition_of_sodium_chlorate.doc
http://shelbychem.weebly.com/uploads/4/5/0/3/4503102/lead_iodide_lab.doc